The LabCoat Guide to Pesticide Mode of Action: Insecticides – Part II
In this fourth article in the LabCoat Guide to Pesticide Mode of Action, I discuss conventional Insecticides and BioInsecticides, and their Biochemical Mode of Activity.
A little about my background
I am a plant scientist with a background in Molecular Plant Biology and Crop Protection. 20 years ago, I worked at Copenhagen University on photosynthetic responses to stress in crops. Subsequently, I worked in Australia on molecular defense mechanisms induced by Phylloxera attack in grapevine.
At that time, biology-based crop protection strategies had not taken off commercially, so I transitioned to conventional (chemical) crop protection R&D at Cheminova, later FMC.
During this period, public opinion, as well as increasing regulatory requirements, gradually closed the door of opportunity for conventional crop protection strategies, while the biological crop protection technology I had contributed to earlier began to reach commercial viability.
From January 2018, I will consolidate 17+ years of industry experience in BioScience R&D management with my academic research background, to provide independent Strategic R&D Management as well as Scientific Development and Regulatory support to AgChem & BioScience organizations developing science-based products.
For more information, visit BIOSCIENCE SOLUTIONS – Strategic R&D Management Consultancy.
***
In the previous article in this series, I discussed the importance of a basic knowledge of insecticide mode of action for determining the optimal use of insecticides, as well as for understanding the underlying mechanisms of insect resistance and insecticide selectivity.
Following a brief classification of insecticide modes of action, I identified insecticides targeting the nervous systems of insects as the main commercial group of relevance today.
A neural template was used to explain the mode of action of insecticides directly impacting the Cholinergic (ACh) synapse. In this article, I discuss additional insecticide modes of action using this template, as well as identify and discuss key classes of BioInsecticides.
Inhibition of the GABA synapse by insecticides
In GABA (Gamma Amino Butyric Acid) neurons, nerve impulses arriving at the axon terminal trigger the release of inhibitory GABA neurotransmitters from neural vesicles. These neurotransmitter molecules are released from the axon terminal and traverse the synaptic cleft before binding to specific receptors on the surface of postsynaptic dendrites.
In contrast to activatory Cholinergic (ACh) synapses, GABBA neurotransmitters are termed inhibitory neurotransmitters, as they reduce or inhibit post-synaptic neural activation.
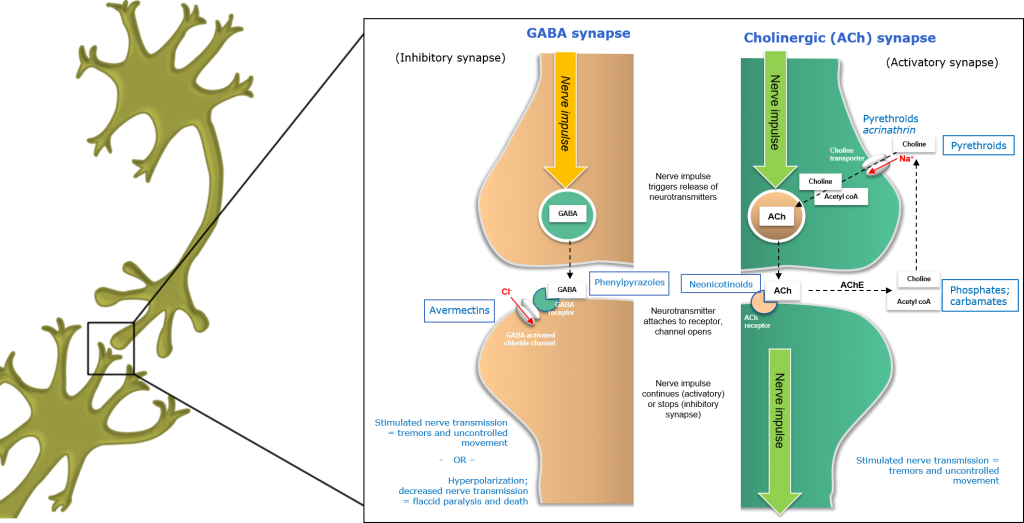
Figure 1: Inhibition of the GABBA synapse in insects.
One class of GABA inhibitors causes hyperstimulation, by blocking inhibitory GABA neurotransmitters and removing the damping effects of GABA, which maintains normal nerve activity. Fipronil is a phenylpyrazole insecticide which binds to the GABA receptor, blocking insect-specific GABA-and glutamate-gated chloride channels and causing neural hyperstimulation similar to that observed for pyrethroids.
In contrast, Avermectins affect insect-specific glutamate-gated chloride channels in GABA neurons, causing an influx of chloride ions (Cl-) into the postsynaptic neuron – leading to hyperpolarisation, paralysis and death.
Neural agonists and antagonists
Insecticides targeting nervous systems may behave as agonists (activating the receptors to which they bind, increasing the inhibitory or excitatory effect of neurotransmitters) such as the Neonicotinoids or Avermectins, or antagonists (binding to receptors without activating them and potentially blocking the activity of agonists), such as Fipronil.
Insecticide specificity
Some insecticides have a high specificity towards insects. For example, it is believed that Fipronil’s insect-specificity is due to its greater affinity for insect GABA receptors, relative to GABA receptors in mammals. Insecticides affecting glutamate-gated chloride channels are specific to insects, as these channels do not exist in mammals. Accordingly, insect-specific insecticides can be considered for use on mammals, for example as flea control products.
In contrast to specific insecticides, other insecticides may target metabolic processes which are common to insects and mammals. As the AChE enzyme is similar in insects and mammals, this group of insecticides is not considered selective, and appropriate care must be exercised in their application.
BioInsecticides
BioInsecticides fall into three major classes:
I) Microbial insecticides
- Entomopathogenic fungi, capable of causing disease in insects (e.g. Beauveria)
- Beneficial nematodes attacking insects (e.g. Steinernema)
- Entomopathogenic viruses (e.g. Cydia pomonella granulovirus)
II) Plant-incorporated protectants
PIPs comprise foreign DNA inserted into crop genetic material (GM crops). Their use may be restricted, for example in certain EU countries. An example is:
- Bacillus thuringiensis, an entomopathogenic disease, functions as an insecticide when its toxin (Bt toxin) is directly incorporated into plants through the use of genetic engineering
III) Biochemical pesticides
Naturally occurring compounds, including fungal and plant extracts that control insects. Examples include:
- Insect pheromones, attracting insects to traps or disrupting mating cycles
- Insecticidal compounds isolated from microorganisms (e.g. Spinosad, or Bt toxin). When Bt toxin is ingested by the insect, insecticidal protein toxins are activated and lead to cell disintegration and insect death. BT toxins may be specific for caterpillars, beetles or flies and have little effect on mammals
- Chitosan derived from fungi or crustacean shells, capable of inducing systemic resistance against insect pests, such as parasitic cyst nematodes
- Plant extracts such as garlic extracts or canola oil, capable of repelling insects, or extracts
- Plant extracts with neural insecticidal activity such as Nicotine, Pyrethrins – isolated from Chrysanthemum plants, or Ryanoids, insecticidal alkaloids which activate ryanodine calcium-channel receptors in insects, leading to paralysis and death
- Plant extracts capable of inhibiting mitochondrial electron transport (respiration) such as Rotenone, an insecticide which may be metabolised in warm-blooded animals (but not e.g. fish)
- Naturally occurring desiccants, such as borate or silica
RNA interference (RNAi) is an emerging technology, whereby RNA molecules capable of disrupting genes conferring tolerance to insecticides may be used to target resistant insects such as Potato Beetles.
In a previous series on BioPesticides, I elucidate some of the factors affecting their efficacy and commercial viability.
In other articles in this series, I focus on herbicide, fungicide and insecticide modes of action, as well as basic physicochemical and formulation concepts related to pesticide activity.
Harry Teicher is the founder of BIOSCIENCE SOLUTIONS and an Authorpreneur, providing organizations with Strategic- and Project Management as well as Development & Communication solutions. Follow him on Linkedin, Twitter and Facebook.