The LabCoat Guide to Pesticide Mode of Action: Pesticide Solubility and Biological Parameters
In this sixth article in the LabCoat Guide to Pesticide Mode of Action, I discuss Pesticide Solubility and Biological Parameters.
A little about my background
I am a plant scientist with a background in Molecular Plant Biology and Crop Protection. 20 years ago, I worked at Copenhagen University on photosynthetic responses to stress in crops. Subsequently, I worked in Australia on molecular defense mechanisms induced by Phylloxera attack in grapevine.
At that time, biology-based crop protection strategies had not taken off commercially, so I transitioned to conventional (chemical) crop protection R&D at Cheminova, later FMC.
During this period, public opinion, as well as increasing regulatory requirements, gradually closed the door of opportunity for conventional crop protection strategies, while the biological crop protection technology I had contributed to earlier began to reach commercial viability.
From January 2018, I will consolidate 17+ years of industry experience in BioScience R&D management with my academic research background, to provide independent Strategic R&D Management as well as Scientific Development and Regulatory support to AgChem & BioScience organizations developing science-based products.
For more information, visit BIOSCIENCE SOLUTIONS – Strategic R&D Management Consultancy.
***
The ability to assess the physicochemical properties of pesticide active ingredients and to apply this knowledge to biological parameters is an important factor in pesticide development.
Depending on its intended use, a pesticide active ingredient (a.i.) may require an element of water solubility (hydrophilicity) to allow it to be transported and distributed within the plant, while having some non-aqueous solubility (lipophilicity), to allow it to permeate across biological membranes.
Lipophilicity (logP)
Lipophilicity describes a molecule’s ability to dissolve in lipophilic (non-aqueous) solutions, allowing permeation through biological membranes.
Lipophilicity is measured as the partition coefficient (Kow) of a given molecule between non-aqueous (octanol, o) and aqueous (water, w) phases, and is expressed as the logarithm of the ratios between these phases (logKow or logP). A logP value of 3 indicates a distribution of one molecule in the aqueous phase for every 1000 molecules in the lipophilic (octanol) phase – the molecule may be considered to be lipophilic. For logP=0, the molecules will be equally partitioned between the two phases, while for logP values < 0 the molecules will be preferentially partitioned in the aqueous phase.
LogP only describes the partition coefficient of neutral (uncharged) molecules. In order to predict pesticide mobility in plants, we need to consider the dissociation constant of the molecule, i.e. the charge of the molecules at physiological pH. In contrast, the distribution coefficient (logD) is measured at different pH levels, and thus takes into account the charge of the active ingredients molecules.
pKa
The acid dissociation constant (Ka) describes the strength of an acid and is typically expressed as pKa (-log10(Ka). The pKa value of a given molecule defines the pH at which it is neutral. At greater pH values, acid groups will be charged, while at lower pH values basic groups will be charged. The number and distribution of charges on a molecule affect its aqueous solubility.
Solubility and uptake
Solubility of the active ingredient is determined by its lipophilicity and dissociation constant. Lipophilic active ingredient molecules may resist solubility in water – the most common medium for agricultural sprays – and typically require formulation with organic solvents or oils.
Once on the insect or leaf surface, the lipophilicity of these molecules allows the rapid uptake of the active ingredient across lipophilic waxy, cuticular and chitin surface barriers, as well as facilitating extracellular movement within biological cell membranes and cell walls.
In contrast, hydrophilic active ingredient molecules are easily solubilized in aqueous spray solutions, while the uptake of these molecules across lipophilic plant leaf barriers often requires the inclusion of surfactants in the final product. Delay or insufficient uptake of the active ingredient may lead to reduced efficacy, wash-off of the active ingredient by rain or UV-degradation of the unprotected molecules.
Translocation
Within the plant, active ingredient molecules may be translocated throughout the plant according to their lipophilicity and dissociation constants. If the active ingredient remains on the plant surface, it is described as having contact activity, whereas if the active ingredient is translocated throughout the plant, it is described as having systemic activity.
Contact pesticides tend to be lipophilic, and are absorbed into their waxy cuticular of plants, where they resist wash off by rain. Contact insecticides are particularly effective against so-called rasping insects, such as mites, which feed on surface cells. Alternatively, lipophilic insecticides may be applied to the soil, where their lipophilicity contributes to reduced leaching of the active ingredients.
Systemic pesticides tend to be hydrophilic, and – depending on the logP and pKA of the active ingredient – translocation of systemic pesticides may take place via the phloem or xylem transport tissues in vascular plants. The phloem transports sap bidirectionally, from existing leaves upward in the plant to new developing tissue, as well as downwards to the roots, to which glucose formed in leaves may be translocated and stored in the form of sucrose. In contrast, the function of the xylem is to transport water unidirectionally from roots to shoots.
Thus, systemic active ingredients transported via the phloem (symplastic transport) may be translocated throughout the plant, including to leaves not otherwise reached by these spray solution, as well as to the roots (of particular relevance for insecticidal active ingredients). Active ingredients transported by the phloem are ingested by sucking insects such as aphids, which preferentially feed on the nutrient-rich phloem. Systemic active ingredients transported via the xylem (apoplastic transport) are translocated “upwards” in the plant, from lower leaves to shoots, but not to the roots.
Solubility and pesticide mobility within plants – the Bromilow Model
The Bromilow model was developed and presented by RH Bromilow et al in the early 1990s, and considers the dissociation coefficient (logP) while taking into account the charge of the active ingredient molecules. LogP and pKa are plotted against each other, and the resulting diagram is a useful model for the prediction of pesticide mobility in plants at physiological pH.
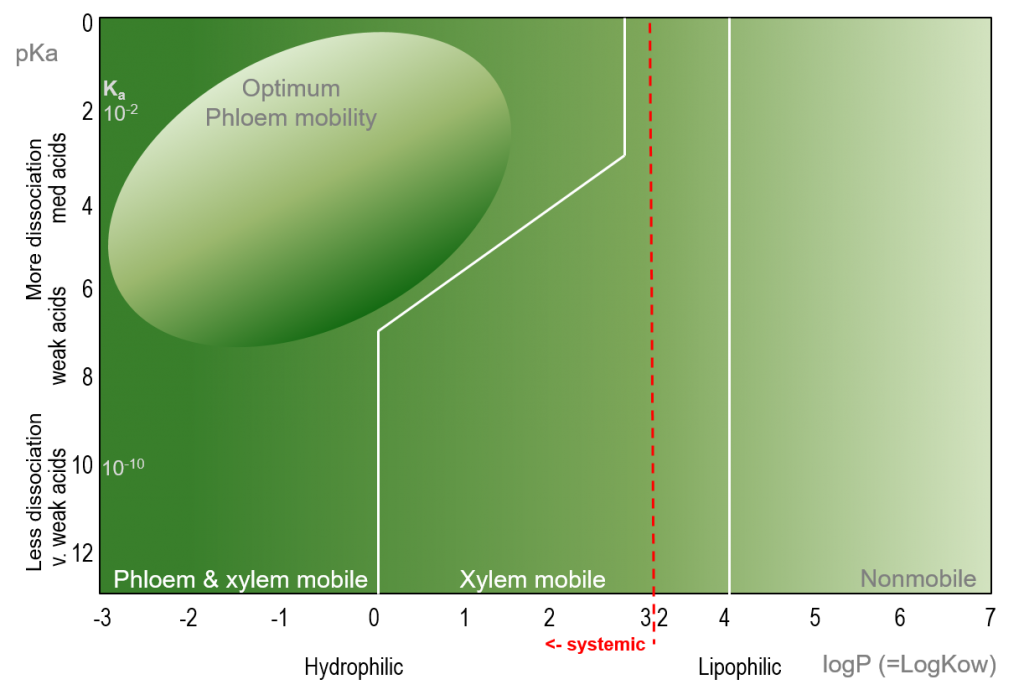
Figure 1. Bromilow model for the prediction of pesticide mobility in plants (degrees of mobility as a function of logP and pKa), adapted from Bromilow et al. (1991).
In the model, pesticide mobility is classified as phloem- & xylem-mobile, xylem mobile or non-mobile. Depending on the logP and pKa of the pesticide molecule, a pesticide may be classified as non-mobile (contact) or mobile (systemic).
Pesticide active ingredients with a logP value of less than 3.2 are considered to be systemic, with hydrophilicity and systemicity increasing with decreasing logP values. Decreasing pKa is associated with increased dissociation of the pesticide active ingredient, with a concomitant increase in plant systemicity.
Application of the Bromilow Model – hydrophilic versus lipophilic herbicides
Glyphosate (GLY) is a non-selective, water-soluble herbicide with a logP of -2.8 and a dissociation constant of about pKa≈2.34 (glyphosate exists as a series of zwitterions, and therefore exists in different ionic states depending on pH), placing it into the optimum phloem mobility category. Glyphosate is readily translocated throughout the plant via the phloem and xylem, moving from sprayed leaves to new shoots, where it inhibits the catalytic function of the EPSP (5-endopyruvylshikimate-3 phosphate synthase) enzyme, thereby inhibiting the formation of aromatic amino acids necessary for plant growth. Glyphosate is not persistent in soils, and does not normally leach to groundwater.
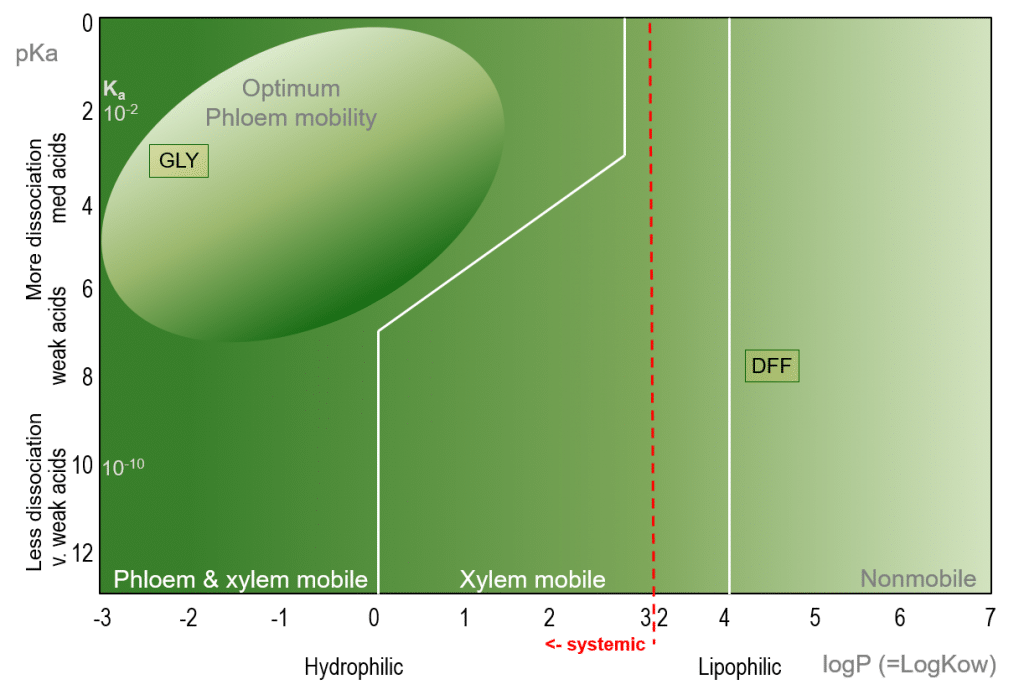
Figure 2. Bromilow model for the prediction of herbicide mobility in plants of Glyphosate (GLY) and Diflufenican (DFF).
In contrast, Diflufenican (DFF) is a lipophilic herbicide (logP=4.2) used to control grasses and broadleaf weeds, with a non-systemic mode of action. DFF is non-mobile within plants, and its herbicidal activity is restricted to the immediate vicinity of the application droplets. DFF is used as a pre-emergent herbicide, and is typically sprayed directly to the soil surface, where it forms a stable, lipophilic herbicide barrier. Germinating weed shoots come into contact with the active ingredient as they break through the soil surface, which causes bleaching by blocking the production of protective plant antioxidants through its inhibitory effect on the enzyme PDS (phytoene desaturase), ultimately leading to death of the seedling through oxidative damage.
Application of the Bromilow Model – herbicide metabolism
When evaluating the translocation of a molecule within a plant, it may be necessary to determine whether the active ingredient is metabolised immediately after uptake. For example, the grass-weed herbicide Fenoxaprop-p-ethyl (FPE; logP=4.58; pkA=0.18) is formulated as an ester, which facilitates its uptake across lipophilic plant barriers. Within the plant, however, the molecule is de-esterified to its toxic form, and the resulting Fenoxaprop-p molecule (FP; logP=1.5 ) exhibits herbicidal activity as well as increased hydrophilicity, facilitating translocation through the plant via both the xylem and phloem.
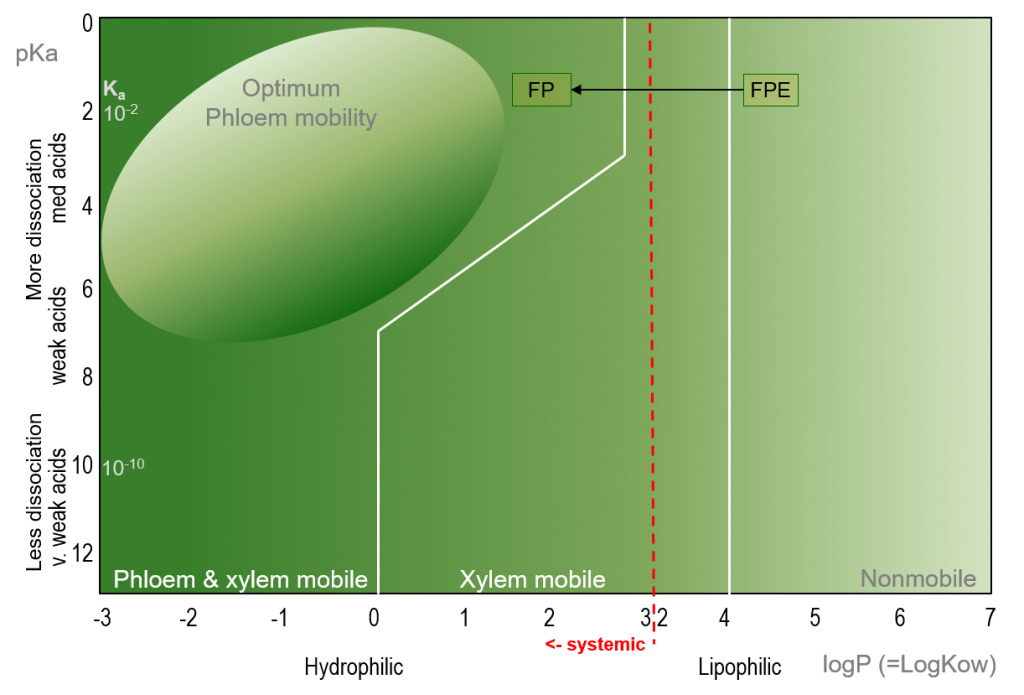
Figure 3. Bromilow model for the prediction of herbicide mobility in plants of Fenoxaprop-p-ethyl (FPE) and Fenoxaprop-p (FP).
Application of the Bromilow Model – insecticide, fungicide and herbicide overview
By inserting herbicide, fungicide and insecticide active ingredients into the Bromilow model, we are able to determine and visualise pesticide mobility for a broad range of active ingredients. In the figure below we see that many common herbicides tend towards xylem and phloem mobility; they are systemic and are therefore typically applied as post-emergent (Post em.) foliar treatments, while some herbicides such as DFF and Pendimethalin (PEN; logP=5.4, pKa=2.8) – an inhibitor of cell division and thus root and shoot growth – are non-systemic, and are applied as a pre-emergence (or early post-emergence) soil applications.
Insecticides may show great systemicity within the plant e.g. Imidacloprid (IMD; logP=0.57) making them ideal to combat sucking pests such as aphids, or non-mobility, e.g. Bifenthrin (BIF; logP=6.6) making them suitable for the control of rasping, surface-feeding pests such as mites and thrips.
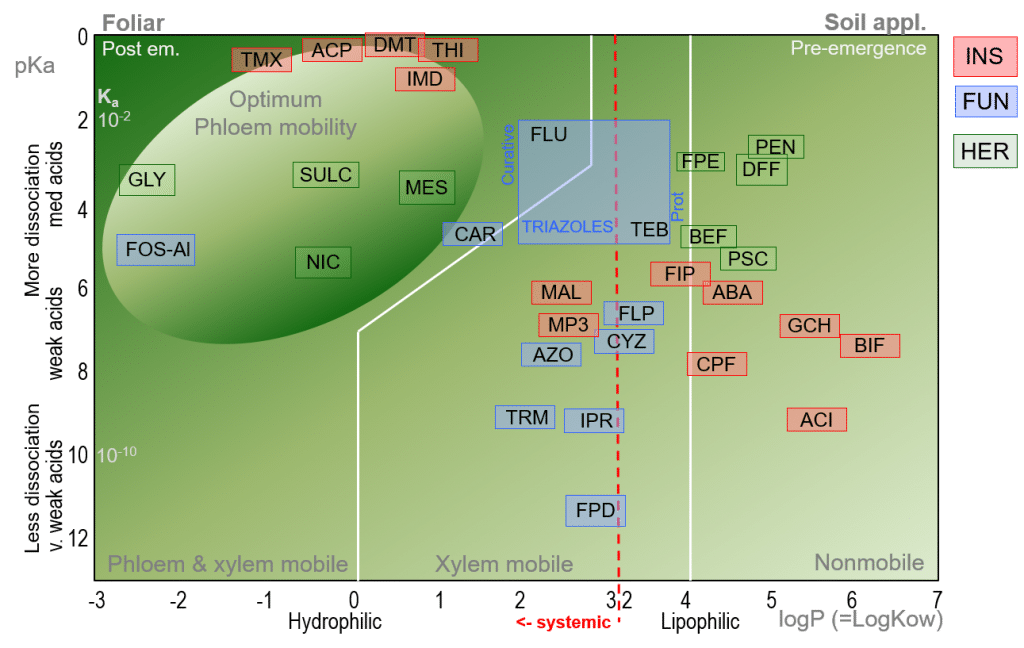
Figure 4. Bromilow model for the prediction of pesticide mobility in plants for common insecticides (INS), fungicides (FUN) and herbicides (HER).
Many common fungicides can be seen to be clustered around the “low” end of the systemicity scale, and are primarily xylem mobile. For triazoles, systemicity may be relatively low e.g. Tebuconazole, TEB; logP=3.7, pKa=5.0) while Flutriafol (FLU; logP=2.3, pKa=2.3) is considered to be the most systemic triazole.
Here, the Bromilow model demonstrates its value for plant physiologists: although the Triazoles lie within a relatively narrow interval in the Bromilow model, their mobility ranges from systemic to relatively non-systemic, and care should be taken before generally recommending Triazole fungicides as curative treatments.
For a fungicide to be considered curative, it is necessary for the active ingredient to be systemically translocated within the plant, to enable it to inhibit (cure) existing fungal hyphal growth within the leaf. In contrast, protective fungicides are generally described as having a contact action, remaining on the surface of the leaf where they are able to protect the plant against the germination of newly-landed fungal spores.
***
Thanks for reading – please help make the BioScience Blog great by liking or sharing this article, making it visible to your network!
Harry Teicher is the founder of BIOSCIENCE SOLUTIONS and an Authorpreneur, providing organizations with Strategic- and Project Management as well as Development & Communication solutions. Follow him on Linkedin, Twitter and Facebook.