The LabCoat Guide to Pesticide Mode of Action: Herbicides & BioHerbicides – Part II
In this second article in the LabCoat Guide to Pesticide Mode of Action, I focus on conventional Herbicides and BioHerbicides, and discuss their Biochemical Mode of Activity and associated plant symptoms.
A little about my background
I am a plant scientist with a background in Molecular Plant Biology and Crop Protection. 20 years ago, I worked at Copenhagen University on photosynthetic responses to stress in crops. Subsequently, I worked in Australia on molecular defense mechanisms induced by Phylloxera attack in grapevine.
At that time, biology-based crop protection strategies had not taken off commercially, so I transitioned to conventional (chemical) crop protection R&D at Cheminova, later FMC.
During this period, public opinion, as well as increasing regulatory requirements, gradually closed the door of opportunity for conventional crop protection strategies, while the biological crop protection technology I had contributed to earlier began to reach commercial viability.
From January 2018, I will consolidate 17+ years of industry experience in BioScience R&D management with my academic research background, to provide independent Strategic R&D Management as well as Scientific Development and Regulatory support to AgChem & BioScience organizations developing science-based products.
For more information, visit BIOSCIENCE SOLUTIONS – Strategic R&D Management Consultancy.
In the previous article in this series, I discussed the importance of a sound knowledge of herbicide mode of action for determining the optimal use of herbicides, as well as for understanding the underlying mechanisms of weed resistance and herbicide selectivity.
The effect of herbicides on enzyme function was explored, as well as the relationship between the activity of herbicides and photosynthesis, and the herbicide-mediated creation of active oxygen radicals.
A photosynthetic template was used to explain the mode of action of herbicides directly impacting photosynthetic electron transport as well as herbicides inhibiting the synthesis of aromatic compounds. In this article, I discuss additional herbicide modes of actions using the photosynthesis template.
Inhibition of Branched-chain Amino Acid Biosynthesis
In addition to the biosynthesis of aromatic amino acids described in the previous article, glucose provides carbon building blocks for all other organic molecules, including branched-chain amino acids. These amino acids are created from the precursor molecule pyruvate, and this process is catalysed by the enzyme ALS (acetolactate synthase). ALS is found in plants and some microorganisms, but not mammals. ALS-inhibitor herbicides prevent the formation of these important amino acids, leading to inhibited plant growth and development.
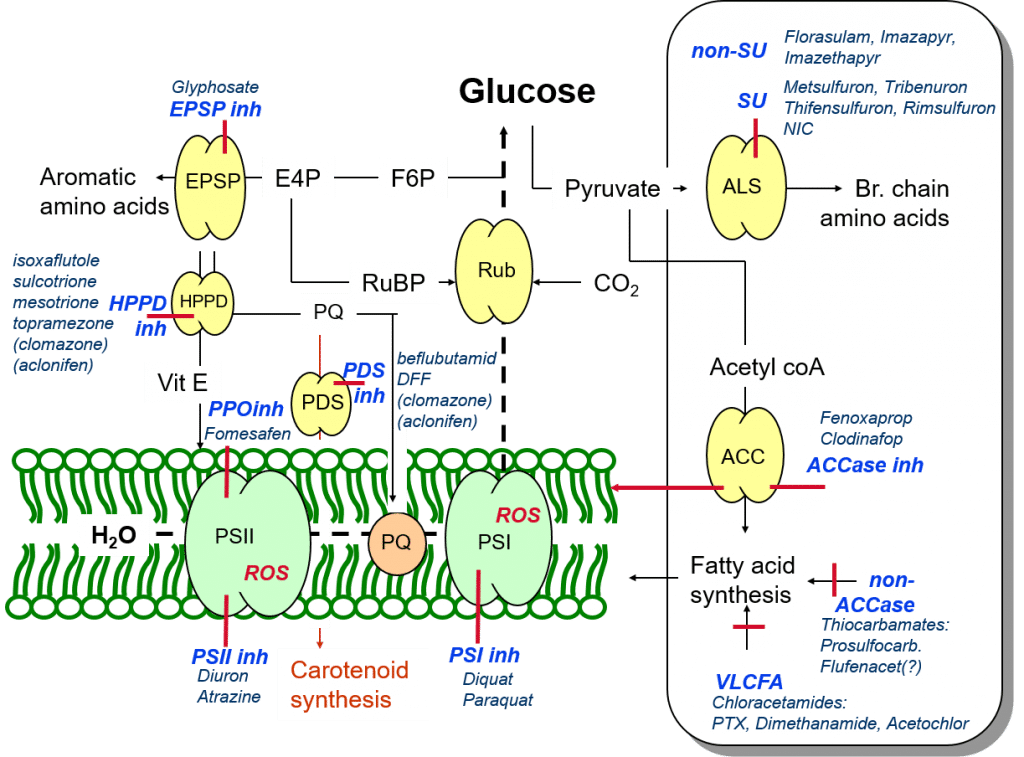
Figure 1: Summary of herbicide modes of action related to the photosynthetic apparatus, amino acid and fatty acid biosynthesis.
Inhibition of Fatty Acid Biosynthesis
In addition to its role in branched-chain amino acid biosynthesis, pyruvate is a precursor for the synthesis of fatty acids, catalysed by the enzyme ACCase (acetyl-CoA carboxylase). Accordingly, ACCase-inhibitor herbicides prevent the formation of new fatty acid-based plant membranes, including thylakoid membranes.
Furthermore, ACCase-enzyme inhibition results in the creation of reactive oxygen species (ROS), while at the same time inhibiting the plant’s ability to maintain and repair membranes damaged by these oxygen radicals. In addition to stunted growth, susceptible plants treated with this group of herbicides typically show the characteristic red discolouration (caused by phytoalexin-based defence responses) of oxidatively stressed plants.
Other herbicide modes of action related to photosynthesis
As described in the previous article, photosynthesis commences with the photolysis of water to oxygen, electrons and protons (hydrogen ions, H+). These protons accumulate on the inner thylakoid membrane space (the thylakoid lumen) giving rise to a proton gradient which is used to drive the creation of ATP by the enzyme ATP synthase. This process of ATP synthesis is termed oxidative phosphorylation. ATP is the universal “molecular unit of currency”, and is a biochemical means of storing energy.
Herbicide uncouplers of oxidative phosphorylation collect protons from the thylakoid lumen and transport them through the thylakoid membrane before releasing them. By lowering the proton gradient, these herbicides reduce the chloroplasts’ ability to produce ATP, resulting in the death of the plant.
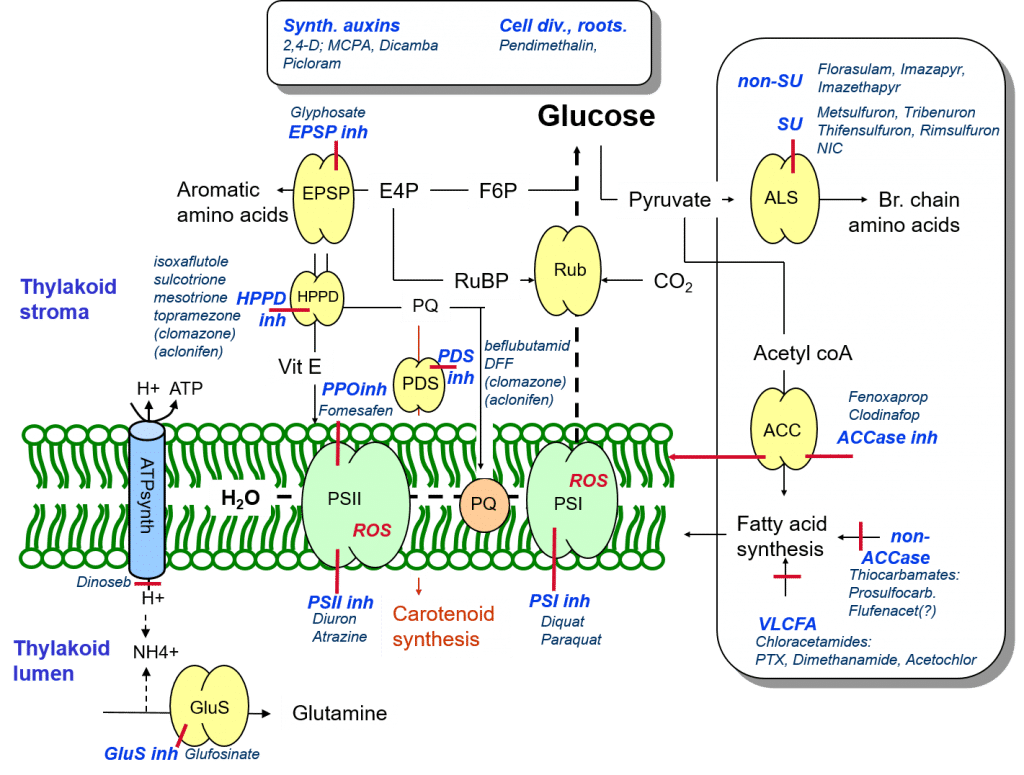
Figure 9: Summary of herbicide modes of action.
Glufosinate is a herbicide that inhibits glutamine synthetase, the enzyme catalysing the production of the amino acid glutamine through the metabolism and detoxification of lumenal ammonia (NH3). This inhibition leads to reduced protein biosynthesis and elevated ammonia and ammonium (NH4+) levels, resulting in plant death. Currently, significant research efforts are being undertaken to identify microorganisms that are capable of producing alternative glutamine synthetase inhibitors.
Herbicides: modes of action not related to photosynthesis
Historically, herbicide development has focused on modes of action related to the photosynthetic electron transport process specific to plants. However, mitochondrial respiratory electron transport processes are evolutionarily related to photosynthetic electron transport, and herbicides affecting photosynthetic electron transport may affect respiration in mammals, insects and microorganisms. This has implications for the environmental impact of this group of pesticides, and is the subject of growing research interest. This evolutionary relationship will be elaborated in a later article.
Not all herbicide mode of action is directly related to the photosynthetic process. Synthetic auxins mimic native plant auxins, plant hormones which regulate plant growth. At high doses, synthetic auxins are toxic to plants, e.g. through the production of ethylene which can inhibit growth, cause leave drop and ultimately plant death. Another group of herbicides not related to the photosynthetic process inhibits cell division and cell elongation in susceptible species.
New sites of action are being explored
Alternative modes of action related to photosynthesis – but not to membrane-bound photosynthetic electron transport – might be sought in related, none-thylakoid processes, such as the Calvin cycle – the carbon fixation pathway whereby CO2 is reduced to glucose. Herbicidally-active metabolites capable of selectively disrupting plant-specific processes such as cellulose biosynthesis are now commercially available.
Bioherbicides and biocontrol
Within the field of biocontrol and biopesticides, mechanisms used by plants to exclude invasive plant species from their immediate vicinity are under close scrutiny. Allelopathy is a mechanism by which plants produce one or more biochemicals (allelochemicals) that affect the growth and survival of other plants. This has, among others, led to the identification of leptospermone, a herbicidal allelochemical synthesised by the Lemon Bottlebrush, and the subsequent development of its commercial chemical analogue, Mesotrione (Callisto).
Microbiological bioherbicide candidates include fungi, bacteria and viruses. The modes of action of fungal bioherbicides include plant cell wall degrading enzymes, photobleaching molecules (macrocidins), secreted disease effectors, phytotoxic molecules such as the diterpene chenopodolin, as well as growth-regulating plant hormones such as indole acetic acid.
Bacterial bioherbicides exhibit phytotoxic activity attributed to specific metabolites as well as the elicitation of plant defence responses such as programmed cell death, through the induction of reactive oxygen species.
Other alternatives to conventional herbicides are currently being developed, including the use of endophytes (microorganisms which colonise plants without causing disease) capable of increasing inoculated crop species’ ability to compete with weeds through the production of phytotoxins in non-host plants. In a previous series on biopesticides, I elucidate some of the factors affecting their efficacy and commercial viability.
In other articles in this series, I focus on herbicide, fungicide and insecticide modes of action, as well as basic physicochemical and formulation concepts related to pesticide activity.
Harry Teicher is the founder of BIOSCIENCE SOLUTIONS and an Authorpreneur, providing organizations with Strategic- and Project Management as well as Development & Communication solutions. Follow him on Linkedin, Twitter and Facebook.